Comparisons of the side effects of the post-operative chemotherapies in esophageal squamous cell carcinoma patients
-
摘要:
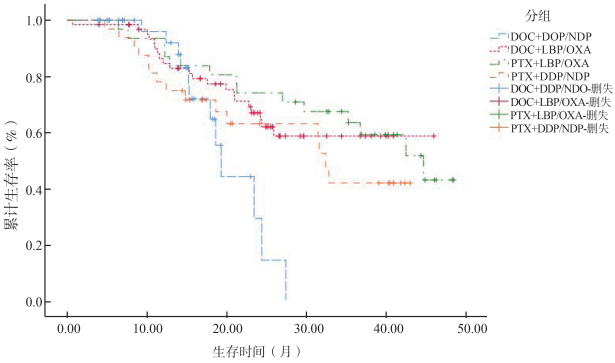
目的 探讨食管鳞癌(esophageal squamous cell carcinoma,ESCC)术后化疗方案与预后生存及副作用的关系。 方法 收集2014年6月―2018年11月18日于福建医科大学附属第一医院、福建省肿瘤医院就诊的ESCC患者病例,根据明确的纳入和排除标准,最终纳入188例。使用Kaplan-Meier法计算累计生存概率,log-rank进行组间差异比较,Cox回归分析生存影响因素,应用多组独立样本秩和检验比较化疗方案组间的副作用,采用非条件Logistic回归分析模型分析化疗方案与ESCC术后副作用的相关性。 结果 化疗方案是ESCC预后的影响因素,其中“多西他赛(docetaxel,DOC)+洛铂(luoplatinum,LBP)/奥沙利铂(oxaliplatin,OXA)”、“紫杉醇(paclitaxel,PTX)+LBP/OXA”化疗方案总生存期均优于“DOC+顺铂(cisplatin,DDP)/奈达铂(nedaplatin,NDP)”组化疗方案;化疗方案与ESCC术后出现的血液/骨髓异常等副作用相关(χ2=11.741,P=0.008),“DOC+LBP/OXA”组较“DOC+DDP/NDP”组化疗方案,会增加白细胞降低发生的风险(OR=4.089,95% CI:1.073~15.585,P=0.039),减少淋巴细胞水平降低发生的风险(OR=0.257,95% CI:0.075~0.878,P=0.030)。 结论 化疗方案是ESCC术后预后生存及副作用的影响因素,“DOC+LBP/OXA”、“PTX+LBP/OXA”两组化疗方案的预后生存情况优于“DOC+DDP/NDP”化疗方案;“DOC+LBP/OXA”化疗方案相对其它方案可降低副作用的发生风险。 Abstract:Objective To investigate the relationship between post-operative chemotherapy, prognosis and side-effects of esophageal squamous cell carcinoma (ESCC). Methods This study enrolled a total of 188 patients with ESCC in the First Affiliated Hospital of Fujian Medical University and Fujian Cancer Hospital from June 2014 to November 18th 2018. The cumulative survival rates were estimated using the Kaplan-Meier method and comparisons were performed using the log-rank test. The Cox proportional hazards model was performed for analysis of the impact factors of survival and the Kruskal-Wallis H test was used to compare side-effects of chemotherapy regimens. The unconditional Logistic regression was performed to analyze the correlation between postoperative chemotherapy regimens and side-effects. Results The chemotherapy regimen was an independent risk factor for poor survival of ESCC patients. Compared with "docetaxel (DOC)+cisplatin (DDP)/nedaplatin (NDP)" group, ESCC patients in "paclitaxel (PTX)+luoplatinum (LBP)/oxaliplatin (OXA)" group and "DOC +LBP/OXA" group had better overall survival. The post-operative chemotherapy regimen was related to blood/bone marrow abnormalities and other side-effects (χ2=11.741, P=0.008). Compared with "DOC+DDP/NDP" group, "DOC+LBP/OXA" group increased the risk of leukocyte reduction (OR=4.089, 95% CI:1.073~15.585, P=0.039) and decreased the risk of lymphocyte reduction (OR=0.257, 95% CI:0.075~0.878, P=0.030). Conclusions The chemotherapy regimen is the impact factor for the prognosis and side-effects of patients with ESCC. The prognosis of "DOC+LBP/OXA" group and "PTX+LBP/OXA" group is better than "DOC+DDP/NDP" group. The "DOC+LBP/OXA" group reduces the risk of side-effects. -
表 1 ESCC患者化疗前的基本情况
Table 1. Basic information of ESCC patients before chemotherapy
变量 化疗方案[n(%)]/[M(P25,P75)] /(x±s) χ2/H值 P值 DOC+DDP/NDP DOC+LBP/OXA PTX+LBP/OXA PTX+DDP/NDP 例数 44 77 33 34 性别 1.431 0.698 女 7(15.9) 16(20.8) 6(18.2) 9(26.5) 男 37(84.1) 61(79.2) 27(81.8) 25(73.5) 年龄(岁) 60.00(54.00, 64.50) 59.00(52.5, 62.5) 61.00(55.00, 65.00) 59.50(54.00, 64.00) 2.337 0.506 白细胞计数(109/L) 6.47(5.25, 7.97) 6.97(5.84, 8.30) 7.40(6.00, 9.20) 6.80(5.80, 8.40) 3.918 0.270 淋巴细胞计数(109/L) 1.60(1.22, 2.09) 1.90(1.58, 2.35) 2.5(2.00, 2.70) 1.80(1.50, 2.20) 22.464 <0.001 红细胞计数(1012/L) 4.41(4.26, 4.55) 4.45(4.09, 4.73) 4.72(4.27, 4.97) 4.60(4.17, 4.93) 9.227 0.026 血红蛋白(g/L) 137.00(129.00, 145.00) 136.00(121.00, 145.00) 142.00(133.00, 150.00) 140.00(129.00, 148.00) 4.428 0.219 白蛋白(g/L) 39.35(38.00, 42.90) 39.65(36.75, 42.35) 39.20(6.30, 40.10) 40.70(36.90, 42.60) 2.694 0.441 谷丙转氨酶(U/L) 15.00(11.00, 21.50) 15.00(11.50, 23.00) 16.00(12.00, 22.00) 16.50(11.00, 23.00) 0.046 0.997 谷草转氨酶(U/L) 21.00(16.00, 24.00) 18.00(16.00, 24.00) 19.00(16.00, 26.00) 20.00(17.00, 26.00) 1.958 0.581 尿素氮(mmol/L) 5.00(4.30, 6.10) 4.69(3.85, 5.52) 5.37(4.28, 6.55) 4.86(4.11, 6.26) 7.099 0.069 肌酐(μmol/L) 74.39±11.92 75.37±15.15 83.00±13.01 82.18±13.15 14.087 0.003 病理分期 6.808 0.078 Ⅰ/Ⅱ 16(42.1%) 22(31.4%) 11(36.7%) 5(14.7%) Ⅲ 22(57.9%) 48(68.6%) 19(63.3%) 29(85.3%) 肿瘤部位 0.618 0.892 上段/中段 28(68.3%) 51(70.8%) 24(72.7%) 22(64.7%) 下段 13(31.7%) 21(29.2%) 9(27.3%) 12(35.3%) 表 2 影响术后化疗患者预后的单因素和多因素分析
Table 2. Univariate and multivariate analysis of prognostic factors in the post-operative chemotherapy in ESCC patients
变量 例数(n) 单因素 多因素 HR(95% CI)值 P值 HR(95% CI)值 P值 年龄(岁) ≤60 101 1.000 1.000 >60 86 0.990(0.665~1.472) 0.959 1.130(0.707~1.808) 0.609 性别 女 38 1.000 1.000 男 150 0.911(0.568~1.462) 0.699 1.259(0.704~2.250) 0.437 淋巴细胞计数(109/L) 0.626(0.460~0.852) 0.003 0.868(0.594~1.268) 0.463 红细胞计数(1012/L) 0.569(0.375~0.863) 0.008 0.860(0.496~1.494) 0.593 肌酐(μmol/L) 0.978(0.963~0.994) 0.006 0.983(0.964~1.003) 0.090 肿瘤部位 上段/中段 125 1.000 1.000 下段 55 0.917(0.579~1.452) 0.711 0.867(0.522~1.439) 0.580 病理分期 Ⅰ/Ⅱ 54 1.000 1.000 Ⅲ 118 0.935(0.613~1.426) 0.755 1.096(0.671~1.790) 0.715 化疗方案 DOC+DDP/NDP 44 1.000 1.000 DOC+LBP/OXA 77 0.256(0.146~0.449) <0.001 0.257(0.130~0.507) <0.001 PTX+LBP/OXA 33 0.047(0.021~0.105) <0.001 0.057(0.023~0.139) <0.001 PTX+DDP/NDP 34 0.127(0.062~0.261) <0.001 0.127(0.056~0.288) <0.001 表 3 化疗方案的组间副作用比较
Table 3. Comparison of side effects between groups of chemotherapy regimens
化疗方案与副作用 分级[n(%)] χ2值 P值 0级 1~级 3~4级 白细胞降低 3.838 0.279 DOC+DDP/NDP 41(93.2) 1(2.3) 2(4.5) DOC+LBP/OXA 61(79.2) 16(20.8) 0(0.0) PTX+LBP/OXA 26(78.8) 7(21.2) 0(0.0) PTX+DDP/NDP 28(82.4) 6(17.6) 0(0.0) 血红蛋白降低 2.540 0.468 DOC+DDP/NDP 37(84.1) 7(15.9) 0(0.0) DOC+LBP/OXA 62(80.5) 15(19.5) 0(0.0) PTX+LBP/OXA 29(87.9) 4(12.1) 0(0.0) PTX+DDP/NDP 25(73.5) 9(26.5) 0(0.0) 淋巴细胞降低 11.741 0.008 DOC+DDP/NDP 34(77.3) 10(22.7) 0(0.0) DOC+LBP/OXA 72(93.5) 5(6.5) 0(0.0) PTX+LBP/OXA 32(97.0) 1(3.0) 0(0.0) PTX+DDP/NDP 32(94.1) 2(5.9) 0(0.0) 疲劳 27.923 <0.001 DOC+DDP/NDP 44(100.0) 0(0.0) 0(0.0) DOC+LBP/OXA 77(100.0) 0(0.0) 0(0.0) PTX+LBP/OXA 33(100.0) 0(0.0) 0(0.0) PTX+DDP/NDP 28(82.4) 6(17.6) 0(0.0) 食欲减退 32.579 <0.001 DOC+DDP/NDP 43(97.7) 1(2.3) 0(0.0) DOC+LBP/OXA 76(98.7) 1(1.3) 0(0.0) PTX+LBP/OXA 32(97.0) 1(3.0) 0(0.0) PTX+DDP/NDP 24(70.6) 10(29.4) 0(0.0) 恶心 14.642 0.002 DOC+DDP/NDP 43(97.7) 1(2.3) 0(0.0) DOC+LBP/OXA 77(100.0) 0(0.0) 0(0.0) PTX+LBP/OXA 32(97.0) 1(3.0) 0(0.0) PTX+DDP/NDP 29(85.3) 5(14.7) 0(0.0) 呕吐 10.026 0.018 DOC+DDP/NDP 44(100.0) 0(0.0) 0(0.0) DOC+LBP/OXA 77(100.0) 0(0.0) 0(0.0) PTX+LBP/OXA 32(97.0) 1(3.0) 0(0.0) PTX+DDP/NDP 31(91.2) 3(8.8) 0(0.0) 谷丙转氨酶升高 0(0.0) 0.319 0.956 DOC+DDP/NDP 41(93.2) 3(6.8) 0(0.0) DOC+LBP/OXA 72(93.5) 5(6.5) 0(0.0) PTX+LBP/OXA 30(90.9) 3(9.1) 0(0.0) PTX+DDP/NDP 32(94.1) 2(5.9) 0(0.0) 谷草转氨酶升高 0.191 0.979 DOC+DDP/NDP 42(95.5) 2(4.5) 0(0.0) DOC+LBP/OXA 74(96.1) 3(3.9) 0(0.0) PTX+LBP/OXA 32(97.0) 1(3.0) 0(0.0) PTX+DDP/NDP 33(97.1) 1(2.9) 0(0.0) 表 4 四组化疗方案对副作用的影响分析
Table 4. Influences of chemotherapy regiments on side effects in the four groups
化疗方案与副作用 分级[n(%)] OR(95% CI)值 P值 0级 ≥1级 白细胞降低 DOC+DDP/NDP 41(93.2) 3(6.8) 1.000 DOC+LBP/OXA 61(79.2) 16(20.8) 4.089(1.073~15.585) 0.039 PTX+LBP/OXA 26(78.8) 7(21.2) 4.366(0.906~21.034) 0.066 PTX+DDP/NDP 28(82.4) 6(17.6) 2.870(0.619~13.315) 0.178 血红蛋白降低 DOC+DDP/NDP 37(84.1) 7(15.9) 1.000 DOC+LBP/OXA 62(80.5) 15(19.5) 1.299(0.460~3.674) 0.621 PTX+LBP/OXA 29(87.9) 4(12.1) 0.827(0.193~3.541) 0.798 PTX+DDP/NDP 25(73.5) 9(26.5) 2.399(0.702~8.203) 0.163 淋巴细胞降低 DOC+DDP/NDP 34(77.3) 10(22.7) 1.000 DOC+LBP/OXA 72(93.5) 5(6.5) 0.257(0.075~0.878) 0.030 PTX+LBP/OXA 32(97.0) 1(3.0) 0.223(0.023~2.170) 0.196 PTX+DDP/NDP 32(94.1) 2(5.9) 0.217(0.038~1.248) 0.087 谷丙转氨酶升高 DOC+DDP/NDP 41(93.2) 3(6.8) 1.000 DOC+LBP/OXA 72(93.5) 5(6.5) 0.809(0.171~3.818) 0.788 PTX+LBP/OXA 30(90.9) 3(9.1) 0.927(0.137~6.287) 0.938 PTX+DDP/NDP 32(94.1) 2(5.9) 0.761(0.102~5.660) 0.789 谷草转氨酶升高 DOC+DDP/NDP 42(95.5) 2(4.5) 1.000 DOC+LBP/OXA 74(96.1) 3(3.9) 0.610(0.088~4.230) 0.617 PTX+LBP/OXA 32(97.0) 1(3.0) 0.379(0.025~5.798) 0.486 PTX+DDP/NDP 33(97.1) 1(2.9) 0.414(0.027~6.329) 0.526 -
[1] Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018:GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6):394-424. DOI: 10.3322/caac.21492. [2] Chen W, Zheng R, Zhang S, et al. Cancer incidence and mortality in China in 2013:an analysis based on urbanization level[J]. Chin J Cancer Res, 2017, 29(1):1-10. DOI: 10.21147/j.issn.1000-9604.2017.01.01. [3] Kanemura T, Makino T, Miyazaki Y, et al. Distribution patterns of metastases in recurrent laryngeal nerve lymph nodes in patients with squamous cell esophageal cancer[J]. Dis Esophagus, 2017, 30(1):1-7. DOI: 10.1111/dote.12527. [4] 相智声, 林征, 刘双, 等.术前血清CA19-9和NSE对食管鳞癌患者预后的影响[J].中华疾病控制杂志, 2019, 23(2):134-139. DOI: 10.16462/j.cnki.zhjbkz.2019.02.003.Xiang ZS, Lin Z, Liu S, et al. Prognostic values of preoperative serum CA199 and NSE on esophageal squamous cell carcinoma patients[J]. Chin J Dis Control Prev, 2019, 23(2):134-139. DOI: 10.16462/j.cnki.zhjbkz.2019.02.003. [5] Lagergren J, Smyth E, Cunningham D, et al. Oesophageal cancer[J]. Lancet, 2017, 390(10110):2383-2396. DOI:10.1016-S0140-6736(17)31462-31469. [6] Chan KKW, Saluja R, Delos Santos K, et al. Neoadjuvant treatments for locally advanced, resectable esophageal cancer:a network meta-analysis[J]. Int J Cancer, 2018, 143(2):430-437. DOI: 10.1002/ijc.31312. [7] Chen Y, Zhang Z, Jiang G, et al. Gross tumor volume is the prognostic factor for squamous cell esophageal cancer patientstreated with definitive radiotherapy[J]. Thorac Dis, 2016, 8(6):1155-1161. DOI: 10.21037/jtd.2016.04.08. [8] National Health Commission of the People's Republic of China. Chinese guidelines for diagnosis and treatment of esophageal carcinoma 2018(English version)[J]. Chin J Cancer Res, 2019, 31(2):223-258. DOI: 10.21147/j.issn.1000-9604.2019.02.01. [9] U.S. Department of Health and Human Services. Cancer therapy evaluation program, common terminology criteria for adverse events, version 4.0[EB/OL]. (2009-05-28)[2020-02-01]. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf. [10] Courrech Staal EF, Aleman BM, van Velthuysen ML, et al. Chemoradiation for esophageal cancer institutional experience with three different regimens[J]. Am J Clin Oncol, 2011, 34(4):343-349. DOI: 10.1097/COC.0b013e3181dbbafe. [11] 刘爱娜, 黄镜, 蔡锐刚, 等. 138例晚期食管癌的化疗疗效和预后因素分析[J].癌症, 2008, 27(4):400-406. DOI: 10.3321/j.issn:1000-467X.2008.04.012.Liu AN, Huang J, Cai RG, et al. Responses of advanced esophageal cancer to chemotherapy and prognostic factors:a report of 138 cases[J]. Chin J Cancer, 2008, 27(4):400-406. DOI: 10.3321/j.issn:1000-467X.2008.04.012. [12] Xi M, Zhang P, Zhang L, et al. Comparing docetaxel plus cisplatin versus fluorouracil plus cisplatin in esophageal squamous cell carcinoma treated with neoadjuvant chemoradiotherapy[J]. Jpn J Clin Oncol, 2017, 47(8):683-689. DOI: 10.1093/jjco/hyx060. [13] 刘鲁平, 张玉星.紫杉醇联合洛铂或顺铂治疗晚期非小细胞肺癌48例比较[J].现代肿瘤医学, 2011, 19(1):78-79. DOI: 10.11569/wcjd.v23.i24.3904.Liu LP, Zhang YX. Comparison of paclitaxel combined with luoplatinum or cisplatin in the treatment of 48 patients with advanced non-small-cell lung carcinoma[J]. J Mod Oncol, 2011, 19(1):78-79. DOI: 10.11569/wcjd.v23.i24.3904. [14] 任超, 周福有.洛铂联合紫杉醇在局部进展期食管癌新辅助治疗中的疗效和安全性观察[J].现代诊断与治疗, 2019, 30(2):248-250. DOI: CNKI:SUN:XDZD.0.2019-02-087.Ren C, Zhou FY. Efficacy and safety observation of luoplatinum combined with Paclitaxel in neoadjuvant treatment of locally advanced esophageal carcinoma[J]. Mod Diagn Treat, 2019, 30(2):248-250. DOI: CNKI:SUN:XDZD.0.2019-02-087. [15] Chen J, Su T, Lin Y, et al. Intensity-modulated radiotherapy combined with paclitaxel and platinum treatment regimens in locally advanced esophageal squamous cell carcinoma[J]. Clin Transl Oncol. 2018, 20(3):411-419. DOI: 10.1007/s12094-017-1734-y. [16] 樊丽萍, 周燕, 赵西侠, 等.奈达铂与洛铂同步放化疗治疗局部晚期宫颈癌的疗效观察[J].中国肿瘤临床与康复, 2017, 24(8):943-946. DOI: 10.13455/j.cnki.cjcor.2017.08.13.Fan LP, Zhou Y, Zhao XY, et al. Efficacy of concurrent chemoradiotherapy with nedaplatin and luoplatinum for locally advanced cervical carcinoma[J]. Chin J Clin Oncol Rehabil, 2017, 24(8):943-946. DOI: 10.13455/j.cnki.cjcor.2017.08.13. [17] 郭亮, 苏克莉, 牛建花, 等.多西他赛单药化疗与联合铂类一线治疗老年晚期非小细胞肺癌的有效性与安全性研究[J].中国社区医师, 2015, 31(12):38-39. DOI: 10.3969/j.issn.1007-614x.2015.12.22.Guo L, Su KL, Niu JH, et al. Effective and safety study of docetaxel single agent chemotherapy and combined with platinum based first-line in the treatment of elderly patients with advanced non-small cell lung cancer[J]. Chin Comm Doc, 2015, 31(12):38-39. DOI: 10.3969/j.issn.1007-614x.2015.12.22. -




 下载:
下载: