Screening signature genes for primary Sjögren′s syndrome based on bioinformatics analysis
-
摘要:
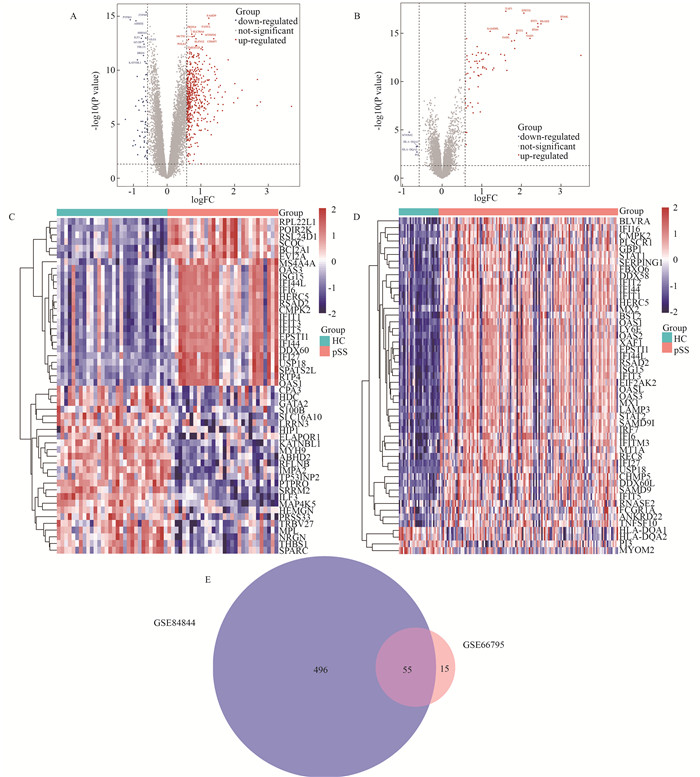
目的 应用生物信息学方法分析确定原发性干燥综合征(primary Sjögren′s syndrome, pSS)患者和健康对照者的特征基因,在转录组学水平上为pSS的发病机制提供思路和理论依据。 方法 从基因表达综合(gene expression opmnibus, GEO)数据库筛选获取pSS患者和健康对照者的芯片数据,数据集GSE84844和GSE66795用于分析获取目标基因,GSE40611用于验证。采用差异分析、加权基因共表达网络分析(weighted gene co-expression network analysis,WGCNA)。利用生物信息学分析方法得到关键基因。通过最小绝对值收敛和选择算子(least absolute shrinkage and selection operator, LASSO)回归获得与pSS发病密切相关的特征基因,受试者工作特征(receiver operating characteristic,ROC)曲线下的面积用来评估特征基因对pSS的诊断价值。 结果 与健康对照者相比,pSS患者共筛选出55个差异表达基因;基因本体(gene ontology, GO)富集分析显示差异表达基因主要参与了抗病毒反应、正调控Ⅰ型干扰素的产生、抗病毒先天免疫反应等生物学过程;京都基因与基因组百科全书(kyoto encyclopedia of genes and geno omes, KEGG)信号通路富集分析发现差异表达基因富集在甲型流感、视黄酸诱导基因蛋白(retinoic acid-inducible gene I, RIG-I)样受体信号通路、坏死性凋亡和乙型肝炎等信号通路;WGCNA联合LASSO回归筛选出4个特征基因,分别为DDX60、EPSTI1、IFI27和IFI44,4个特征基因在验证数据集GSE40611中曲面下面积分别为0.807、0.866、0.804和0.892。 结论 DDX60、EPSTI1、IFI27和IFI44是pSS具有诊断意义的特征基因,能够为更深入地探索原发性干燥综合征的发生发展机制提供理论依据。 -
关键词:
- 原发性干燥综合征 /
- 生物信息学分析 /
- 差异表达基因 /
- 加权基因共表达网络分析 /
- 最小绝对值收敛和选择算子
Abstract:Objective This study employed bioinformatics methods to identify signature genes in patients with primary Sjögren′s syndrome (pSS) compared to healthy controls, offering insights and a theoretical foundation for exploring the pathogenesis of pSS at the transcriptome level. Methods The microarray data containing the information of pSS patients and healthy controls was screened and obtained from the GEO database, with data sets GSE84844 and GSE66795 used to analyze and obtain target genes and GSE40611 used for validation. Differential analysis, weighted gene co-expression network analysis (WGCNA) and other bioinformatics analyses were used to obtain hub genes. The least absolute shrinkage and selection operator (LASSO) regression was used to obtain signature genes closely related to the pathogenesis of pSS, and the area under the receiver operating characteristic (ROC) curve was used to evaluate the diagnostic value of signature genes for pSS. Results Compared with healthy controls, pSS patients had 55 differential expression genes. GO enrichment analysis showed that DEGs were mainly involved in biological processes such as defense response to virus, positive regulation of type Ⅰ interferon production and antiviral innate immune response. Furthermore, KEGG signal pathway enrichment analysis found that DEGs were enriched in influenza A, RIG-I-like receptor signaling pathway, necroptosis and hepatitis B. Four signature genes, DDX60, EPSTI1, IFI27 and IFI44, were screened by WGCNA combined with LASSO regression, and their AUC values in the validation data set were 0.807, 0.866, 0.804 and 0.892, respectively. Conclusions The signature genes DDX60, EPSTI1, IFI27, and IFI44 hold significant diagnostic value for pSS, providing a theoretical foundation for further exploration of the pathogenesis and progression mechanisms of pSS. -
图 4 WGCNA识别关键模块与模块核心基因
A:样本树状和性状图。B:软阈值筛选图。C:基因聚类图。D:模块-特征相关性热图。E:绿松石模块的模块成员和基因重要性之间的相关性图。
Figure 4. WGCNA identified key module and core genes
A: Sample dendrogram and trait heat map. B: Soft thresholding filter diagram. C: Gene cluster diagram. D: Module-trait relationships heat map. E: Gene significance and module membership in turquoise module.
图 6 LASSO回归分析结果
A:确定λ的值。2条虚线表示两个特殊的λ值:lambda.min和lambda.lse(左、右)。B:λ变化时18个关键基因的系数变化情况。上横坐标表示具有非零系数的基因的数量。
Figure 6. Results of LASSO regression analysis
A: Determining the value of λ. Two dotted lines represented two special λ value: lambda.min and lambda.lse(left, right). B: 18 hub genes′ coefficient variation when λ changed. The abscissa above represented the number of variables with non-zero coefficients.
图 7 基因表达箱线图
A-D:DDX60,EPSTI1,IFI27和IFI44在测试数据集中的表达情况。E-H:DDX60,EPSTI1,IFI27和IFI44在验证数据集中的表达情况。
a表示P < 0.001; b表示P < 0.01。Figure 7. Boxplots of gene expression
A-D: Expression of DDX60, EPSTI1, IFI27 and IFI44 in test data set. E-H: Expression of DDX60, EPSTI1, IFI27 and IFI44 in validation data set.
a P < 0.001; b P < 0.01.图 8 特征基因的ROC曲线
ROC:受试者工作特征。
A-D:DDX60,EPSTI1,IFI27和IFI44在测试数据集中的ROC曲线。E-H:DDX60,EPSTI1,IFI27和IFI44在验证数据集中的ROC曲线。Figure 8. ROC curve of feature genes
ROC: receiver operating characteristic
A-D: ROC curve of DDX60, EPSTI1, IFI27 and, IFI44 in test data set. E-H: ROC curve of DDX60, EPSTI1, IFI27 and IFI44 in validation data set.表 1 pSS芯片信息
Table 1. pSS chip information
数据集Data set 平台Platform 样本数目Sample number 样本类型Sample type GSE66795 GPL10558 Illumina HumanHT-12 V4.0 expression beadchip pSS∶Control=131∶29 Whole blood GSE84844 GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array pSS∶Control=30∶30 Whole blood GSE40611 GPL570 [HG-U133_Plus_2] Affymetrix Human Genome U133 Plus 2.0 Array pSS∶Control=17∶18 Parotid gland 表 2 差异表达基因(部分)
Table 2. Differential expression genes (Partial)
序号Serial number 基因名称Gene name Log2差异倍数Log2(Fold Change) P值value 校正后的P值Adjusted P value DEGs_GSE84844 1 IFI27 3.689 <0.001 <0.001 2 IFI44L 2.768 <0.001 <0.001 3 F13A1 -0.580 <0.001 <0.001 4 MYADM -0.580 <0.001 <0.001 DEGs_GSE66795 5 IFI27 3.503 <0.001 <0.001 6 IFI44L 3.014 <0.001 <0.001 7 HLA-DQA1 -0.623 <0.001 0.015 8 PI3 -0.636 0.002 0.038 -
[1] Francois H, Mariette X. Renal involvement in primary Sjögren's syndrome[J]. Nat Rev Nephrol, 2016, 12(2): 82-93. DOI: 10.1038/nrneph.2015.174. [2] Kassan SS, Moutsopoulos HM. Clinical manifestations and early diagnosis of Sjögren's syndrome[J]. Arch Intern Med, 2004, 164(12): 1275-1284. DOI: 10.1001/archinte.164.12.1275. [3] Odani T, Chiorini JA. Targeting primary Sjögren's syndrome[J]. Mod Rheumatol, 2019, 29(1): 70-86. DOI: 10.1080/14397595.2018.1546268. [4] 黄旦, 刘健, 汪元, 等. 中医药治疗干燥综合征合并腺体外器官受累研究进展[J]. 中医药临床杂志, 2022, 34(9): 1759-1762. DOI: 10.16448/j.cjtcm.2022.0942.Huang D, Liu J, Wang Y, et al. Research progress of Chinese medicine treatment of primary Sjögren's syndrome with involvement extra-glandular organ[J]. Clin J Tradit Chin Med, 2022, 34(9): 1759-1762. DOI: 10.16448/j.cjtcm.2022.0942. [5] 邵勤, 吴斌. 原发性干燥综合征的治疗进展[J]. 中国免疫学杂志, 2018, 34(1): 144-148, 157. DOI: 10.3969/j.issn.1000-484X.2018.01.029.Shao Q, Wu B. Treatment progress of primary Sjögren's syndrome[J]. Chin J Immunol, 2018, 34(1): 144-148, 157. DOI: 10.3969/j.issn.1000-484X.2018.01.029. [6] Tasaki S, Suzuki K, Nishikawa A, et al. Multiomic disease signatures converge to cytotoxic CD8 T cells in primary Sjögren's syndrome[J]. Ann Rheum Dis, 2017, 76(8): 1458-1466. DOI: 10.1136/annrheumdis-2016-210788. [7] James K, Al-Ali S, Tarn J, et al. A transcriptional signature of fatigue derived from patients with primary Sjögren's syndrome[J]. PLoS One, 2015, 10(12): e0143970. DOI: 10.1371/journal.pone.0143970. [8] Horvath S, Nazmul-Hossain AN, Pollard RP, et al. Systems analysis of primary Sjögren's syndrome pathogenesis in salivary glands identifies shared pathways in human and a mouse model[J]. Arthritis Res Ther, 2012, 14(6): R238. DOI: 10.1186/ar4081. [9] Zhou Y, Zhou B, Pache L, et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets[J]. Nat Commun, 2019, 10(1): 1523. DOI: 10.1038/s41467-019-09234-6. [10] 张丽宁, 陈嘉琪, 胡琪, 等. 不同舌象原发性干燥综合征患者的免疫炎症特点[J]. 世界中西医结合杂志, 2021, 16(9): 1710-1713. DOI: 10.13935/j.cnki.sjzx.210931.Zhang LN, Chen JQ, Hu Q, et al. Immuno-inflammatory characteristics of primary Sjögren's syndrome patients with different tongue appearances[J]. World J of Integr Tradit West Medi, 2021, 16(9): 1710-1713. DOI: 10.13935/j.cnki.sjzx.210931. [11] 唐小利, 邓红波, 秦悦思, 等. 1例儿童原发性干燥综合征中医诊疗体会[J]. 四川中医, 2022, 40(5): 55-58. https://www.cnki.com.cn/Article/CJFDTOTAL-SCZY202205017.htmTang XL, Deng HB, Qin YS, et al. Traditional Chinese medicine diagnosis and treatment experience for 1 case of primary Sjögren's syndrome in children[J]. Journal of Sichuan of Traditional Chinese Medicine, 2022, 40(5): 55-58. https://www.cnki.com.cn/Article/CJFDTOTAL-SCZY202205017.htm [12] Seror R, Nocturne G, Mariette X. Current and future therapies for primary Sjögren's syndrome[J]. Nat Rev Rheumatol, 2021, 17(8): 475-486. DOI: 10.1038/s41584-021-00634-x. [13] 杨丽, 付萍. 干燥综合征与病毒感染的关系研究进展[J]. 医学综述, 2010, 16(11): 1712-1714. https://www.cnki.com.cn/Article/CJFDTOTAL-YXZS201011041.htmYang L, Fu P. Advances in relations between Sjögren's syndrome and viruses[J]. Medical Recapitulate, 2010, 16(11): 1712-1714. https://www.cnki.com.cn/Article/CJFDTOTAL-YXZS201011041.htm [14] 戴逸君. 细胞因子网络与原发性干燥综合征机制的研究进展[J]. 创伤与急诊电子杂志, 2017, 5(2): 87-93. DOI: 10.16746/j.cnki.11-9332/r.2017.02.010.Dai YJ. Research progress of cytokine network and mechanism of primary Sjögren's syndrome[J]. J Trauma Emerg Electron Version), 2017, 5(2): 87-93. DOI: 10.16746/j.cnki.11-9332/r.2017.02.010. [15] Platanias LC. Mechanisms of type-Ⅰ- and type-Ⅱ-interferon-mediated signalling[J]. Nat Rev Immunol, 2005, 5(5): 375-386. DOI: 10.1038/nri1604. [16] Sun L, Wu J, Du F, et al. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type Ⅰ interferon pathway[J]. Science, 2013, 339(6121): 786-791. DOI: 10.1126/science.1232458. [17] Crow MK, Olferiev M, Kirou KA. Type Ⅰ Interferons in autoimmune disease[J]. Annu Rev Pathol, 2019, 14: 369-393. DOI: 10.1146/annurev-pathol-020117-043952. [18] Meurs E, Chong K, Galabru J, et al. Molecular cloning and characterization of the human double-stranded RNA-activated protein kinase induced by interferon[J]. Cell, 1990, 62(2): 379-390. DOI: 10.1016/0092-8674(90)90374-n. [19] Lee AJ, Ashkar AA. The dual nature of type Ⅰ and type Ⅱ interferons[J]. Front Immunol, 2018, 9: 2061. DOI: 10.3389/fimmu.2018.02061. [20] Imgenberg-Kreuz J, Sandling JK, Almlof JC, et al. Genome-wide DNA methylation analysis in multiple tissues in primary Sjögren's syndrome reveals regulatory effects at interferon-induced genes[J]. Ann Rheum Dis, 2016, 75(11): 2029-2036. DOI: 10.1136/annrheumdis-2015-208659. [21] 王彦焱, 凌彬, 罗莉, 等. 原发性舍格伦综合征患者唇腺组织中IFN-α的表达[J]. 临床和实验医学杂志, 2013, 12(11): 817-818, 821. https://www.cnki.com.cn/Article/CJFDTOTAL-SYLC201311000.htmWang YY, Ling B, Luo L, et al. Expression of interferon-alpha in primary Sjögren's syndrome[J]. Journal of Clinical and Experimental Medicine, 2013, 12(11): 817-818, 821. https://www.cnki.com.cn/Article/CJFDTOTAL-SYLC201311000.htm [22] 郑凌艳, 张志愿, 俞创奇, 等. IFN-α在原发性舍格伦综合征患者外周血中的表达[J]. 上海口腔医学, 2009, 18(2): 121-126. https://www.cnki.com.cn/Article/CJFDTOTAL-SHKY200902008.htmZheng LY, Zhang ZZ, Yu CQ, et al. The expression of interferon-α in peripheral blood of patients with primary Sjögren's syndrome[J]. Shanghai J Stomatol, 2009, 18(2): 121-126. https://www.cnki.com.cn/Article/CJFDTOTAL-SHKY200902008.htm [23] Ferraccioli GF, Salaffi F, de Vita S, et al. Interferon alpha-2 (IFN alpha 2) increases lacrimal and salivary function in Sjögren's syndrome patients. Preliminary results of an open pilot trial versus OH-chloroquine[J]. Clin Exp Rheumatol, 1996, 14(4): 367-371. [24] Ship JA, Fox PC, Michalek JE, et al. Treatment of primary Sjögren's syndrome with low-dose natural human interferon-alpha administered by the oral mucosal route: a phase Ⅱ clinical trial. IFN Protocol Study Group[J]. J Interferon Cytokine Res, 1999, 19(8): 943-951. DOI: 10.1089/107999099313497. [25] Lin Y, Yao X, Yan M, et al. Integrated analysis of transcriptomics to identify hub genes in primary Sjögren's syndrome[J]. Oral Dis, 2022, 28(7): 1831-1845. DOI: 10.1111/odi.13943. [26] Grunvogel O, Esser-Nobis K, Reustle A, et al. DDX60L Is an interferon-stimulated gene product restricting hepatitis C virus replication in cell culture[J]. J Virol, 2015, 89(20): 10548-10568. DOI: 10.1128/JVI.01297-15. [27] Li T, Lu H, Shen C, et al. Identification of epithelial stromal interaction 1 as a novel effector downstream of Kruppel-like factor 8 in breast cancer invasion and metastasis[J]. Oncogene, 2014, 33(39): 4746-4755. DOI: 10.1038/onc.2013.415. [28] 孙金磊. EPSTI1促进原发性干燥综合征B细胞异常活化的机制研究[D]. 北京: 北京协和医学院, 2018.Sun JL. Analysis of differentially expressed genes in primary Sjögren's syndrome by RNA sequencing[D]. Beijing: Peking Union Medical College, 2018. [29] Wang H, Qiu X, Lin S, et al. Knockdown of IFI27 inhibits cell proliferation and invasion in oral squamous cell carcinoma[J]. World J Surg Oncol. 2018, 16(1): 64. DOI: 10.1186/s12957-018-1371-0. [30] Wildenberg ME, van Helden-Meeuwsen CG, van de Merwe JP, et al. systemic increase in type Ⅰ interferon activity in sjögren's syndrome: a putative role for plasmacytoid dendritic cells[J]. Eur J Immunol, 2008, 38(7): 2024-2033. DOI: 10.1002/eji.200738008. [31] Nzeusseu Toukap A, Galant C, Theate I, et al. Identification of distinct gene expression profiles in the synovium of patients with systemic lupus erythematosus[J]. Arthritis Rheum, 2007, 56(5): 1579-1588. DOI: 10.1002/art.22578. [32] Honda Y, Kondo J, Maeda T, et al. Isolation and purification of a non-A, non-B hepatitis-associated microtubular aggregates protein[J]. J Gen Virol, 1990, 71(Pt 9): 1999-2004. DOI: 10.1099/0022-1317-71-9-1999. [33] 李彬彬, 郑燕, 郏雁飞, 等. IFI44的研究现状[J]. 山东科学, 2008, (3): 32-37. https://www.cnki.com.cn/Article/CJFDTOTAL-SDKX200803008.htmLin BB, Zheng Y, Jia YF, et al. A survey of interferon-inducible-protein 44[J]. Shandong Science, 2008, (3): 32-37. https://www.cnki.com.cn/Article/CJFDTOTAL-SDKX200803008.htm [34] 张健. DDX60在肝癌中的表达及功能研究[D]. 广州: 南方医科大学, 2019.Zhang J. The expression and function of DDX60 in hepatocellular carcinoma[D]. Guangzhou: Southern Medical University, 2019. [35] Nishikawa A, Suzuki K, Kassai Y, et al. Identification of definitive serum biomarkers associated with disease activity in primary Sjögren's syndrome[J]. Arthritis Res Ther, 2016, 18(1): 106. DOI: 10.1186/s13075-016-1006-1. -
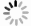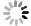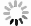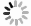




 下载:
下载: